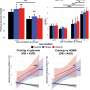
A common cholesterol drug may weaken ovarian cancer’s hidden shield
Ascites—the buildup of liquid in the belly—may be doing more than causing discomfort. A Duke University School of Medicine study finds this fluid helps cancer cells survive and spread—and that a decades-old cholesterol drug may […]